During the second part of today’s seminar, PhD student Ruiyang Wang discussed the results of his recent laboratory experiments on the dissolution of capillary-trapped CO2 in the subsurface.
As CO2 is captured and injected into an aquifer, some of it migrates towards topographic highs within the aquifer, while some is trapped in the pore spaces of the rock by capillary forces. This leads to zones of porous rock where individual pore spaces contain capillary-trapped CO2. Understanding the fate of this trapped CO2 is essential to make sure that the carbon is safely stored underground.
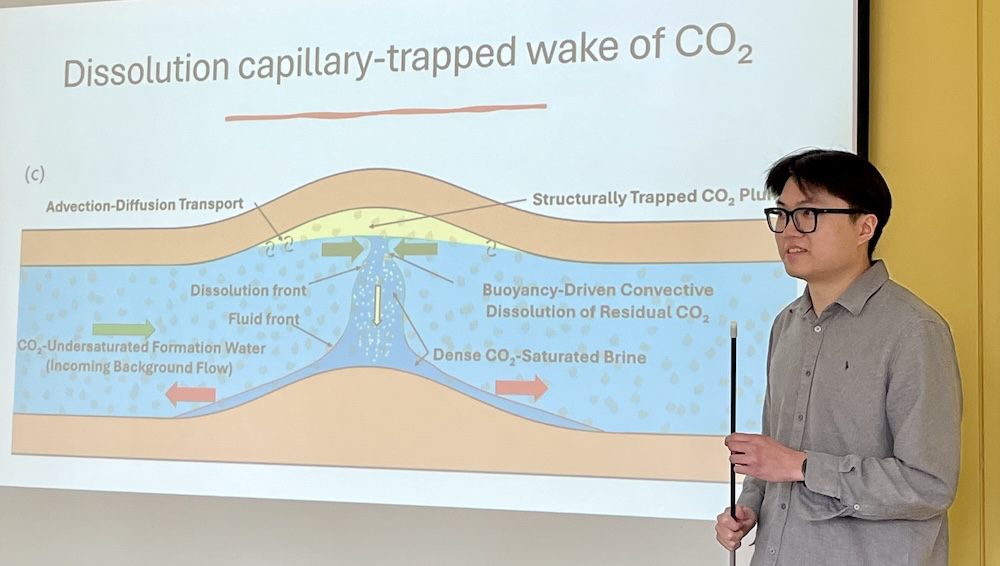
Previous work carried out at the Institute investigated the instabilities which occur when a region of the aquifer containing trapped CO2 overlays a lower region containing unsaturated formation water.
Today, Ruyang is exploring the effects of a different distribution of the trapped CO2 in the aquifer. His experiments show that when the aquifer contains a region with trapped CO2 adjacent to a region without trapped CO2, then a completely different flow develops over time, with relatively dense, CO2-saturated water slumping and displacing the surrounding formation water. As a result of this flow, some of the formation water is recirculated into the portion of the aquifer which contains the trapped CO2, and this leads to further dissolution of the CO2 over time. Using a combination of dye and salt powders, Ruyang’s small-scale analogue laboratory experiments can visualise both the water flow and the progress of the dissolution reaction in real time.
Ruyang’s experimental work is ongoing, and we look forward to hearing more about it very soon!