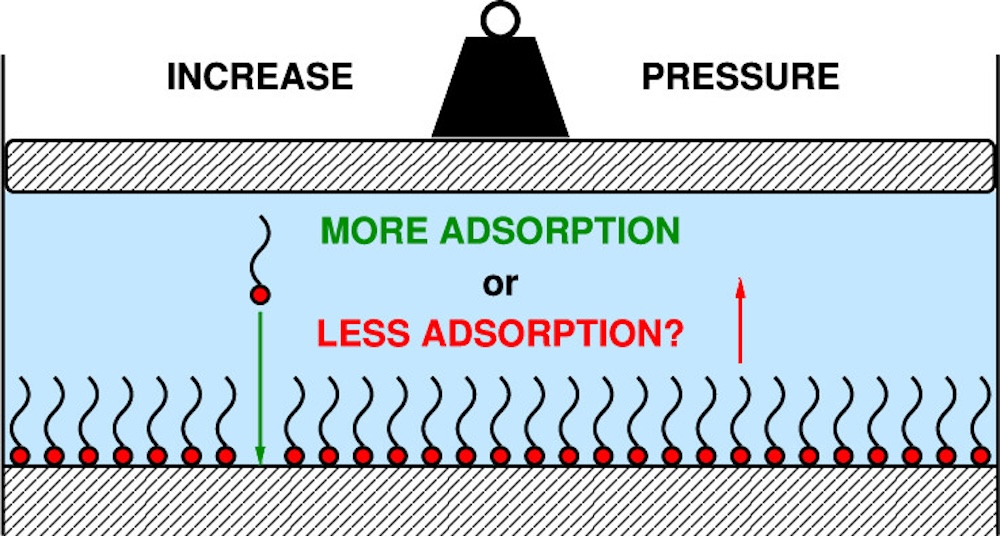
The effect of pressure on gas-phase adsorption on a solid surface has been well-known since the time of Irving Langmuir, but the situation is less clear for the adsorption of a solute from a liquid solution at high pressure. This could be important in solutions of surface-active molecules for applications under high pressure, such as lubricants in engines, motors, and wind turbines in which gigapascal pressures are possible, and the adsorption of additives on moving parts or dispersed particles is linked to performance.
Pressure affects the chemical potentials of the solute and solvent in both the liquid solution and the adsorbed layer. A new article recently published in Langmuir and co-authored by Professor Stuart Clarke explores the effect on liquid-solution thermodynamics, and shows that the volume of mixing of the solution, and its gradient with respect to the solute mole fraction, strongly influence whether adsorption increases or decreases with an increase in pressure.
Dilute solutions with positive (negative) volumes of mixing show increased (decreased) adsorption. In the article, concrete predictions are made using a Langmuir-type model expressed as surface coverage as a function of solute mole fraction. Key thermodynamic relationships are illustrated with molecular-dynamics simulations of a simple model of non-ideal solutions, and numerical estimates of the pressure effect are given. Finally, volume-of-mixing data for real solutions are analysed, and it is shown that gigapascal pressures are enough to change the adsorption constant by at least an order of magnitude.